Quick, Compliant Turnaround of Vital Temperature Sensitive Medicinal Products in Kenya
When it comes to delivering essential medication, time and precision are critical. Delays in the supply chain can directly impact patient outcomes, making speed, accuracy, and compliance non-negotiable. Across our global network, our teams are trusted to handle complex, time-sensitive pharmaceutical operations while maintaining the highest quality standards. This was highlighted by a recent request for our Oximio team in Kenya which showcased streamlined and flawless execution in action.
The Challenge
The Kenyan team received a significant inbound, bulk shipment of Investigational Medicinal Products (IMPs) as part of an urgent CRO order. The shipment required rapid turnaround while ensuring strict adherence to temperature control, quality, and regulatory requirements.
The scope of work included:
- Full unloading of the inbound shipment from the delivery truck
- Detailed checking and verification
- Careful unpacking and preparation readiness for onward redistribution
- Continuous handling within a 2–8°C temperature-controlled environment
Given the volume and critical nature of the products, the task demanded close coordination, attention to detail, and careful processing.
The Solution
Our Kenyan team mobilised quickly, working with focus and precision to successfully complete the entire process in under two working days. All activities were carried out within the designated temperature-controlled specification, ensuring full compliance with quality and regulatory standards throughout the operation.
Despite the demanding nature of the shipment, the team maintained accuracy at every step, from verification through to readiness for onward distribution, demonstrating strong operational discipline and teamwork.
The Outcome
- Large, bulk shipments of IMPs processed efficiently and compliantly
- End-to-end handling completed in under two working days
- 100% adherence to 2–8°C temperature requirements
- IMPs ready for onward distribution
Key Takeaway
This operation highlights the strength of our Kenya team and our broader global capabilities: the ability to move vital medicinal products quickly, safely, and reliably, even under demanding conditions. Their dedication, coordination, and commitment to excellence ensured that critical medications ready for their continued journey without delay.
Clinical Trial Supply Chain Solutions in Africa
With depots in Kenya and South Africa, and a qualified and experienced team on the ground, Oximio can provide tailor-made solutions to support even the most complex or demanding clinical trial requirements.
For further information on our African and global capabilities, visit our website or contact us to discuss your supply chain needs.
Oximio Kenya’s Rapid Response to Marburg Virus Outbreak in Ethiopia
When infectious disease outbreaks emerge, speed, precision, and trust in the supply chain can make a life-saving difference. In late 2025, as Ethiopia faced its first-ever outbreak of Marburg virus disease (MVD), our teams were called upon to act quickly. What followed was a powerful example of how local expertise, global capabilities, and rapid execution can support clinical and public health responses in the most challenging circumstances.
Background: Understanding the Marburg Virus
Marburg virus disease (MVD), formerly known as Marburg haemorrhagic fever, is a rare but severe illness with a high fatality rate. On average, MVD claims around 50% of those infected, with historical outbreaks showing fatality rates ranging from 24% to as high as 88%.
The virus spreads rapidly and can overwhelm healthcare systems if not controlled early. While there are currently no approved vaccines or antiviral treatments, early supportive care, such as rehydration and symptom management, has been shown to significantly improve survival. Community engagement, rapid diagnostics, and well-coordinated clinical response efforts are all essential to containing outbreaks and limiting their impact.
On 14 November 2025, the Ethiopia Ministry of Health officially reported the country’s first Marburg outbreak, triggering an urgent international response.
The Challenge: A Time-Critical Request
In the immediate aftermath of the announcement, Oximio was contacted by the African Disease Division of a large Contract Research Organisation (CRO). They required an urgent delivery of ancillary supplies to support outbreak control efforts. Time was critical. Any delay could hinder frontline response activities during a fast-moving public health emergency.
The responsibility fell to our Kenya-based team, who were tasked with sourcing, preparing, and mobilising the required supplies in the shortest possible timeframe, all whilst under intense pressure and with no margin for error.
Our Response: Agility in Action
Despite the urgency, our teams rose to the challenge. Through seamless coordination, decisive action, and deep regional expertise, the order was completed in under 48 hours and made ready for shipment.
In an outbreak response scenario, this speed is not just impressive, it’s essential. The successful execution demonstrated our ability to operate with agility while maintaining the high standards required in clinical trial and public health supply chains.
The client later shared their sincere appreciation for the exceptional responsiveness and service delivered, reinforcing the trust placed in our teams when it matters most.
Conclusion: Trusted Support When It Matters Most
This case highlights our commitment to supporting clinical trials and public health initiatives across Africa, even in the most demanding circumstances. From rapid mobilisation to reliable execution, our teams continue to demonstrate collaboration, resilience, and dedication to our mission.
To find out how our African-based operations can support your clinical trial and outbreak response needs, please visit our website or contact us to discuss your requirements. We’d be happy to help.
Supporting Customer Growth Across Sub-Saharan Africa
Faced with a growing client base and increased demand for their high-end medical devices and ancillaries, a South African surgical ophthalmic distributor, reached out to Oximio for support in satisfying their own customers’ needs.
The Challenge
Supporting eye care services across the region, they faced several operational and regulatory challenges as they worked to expand their presence, particularly in Kenya and surrounding countries. These included:
- Cross-border logistics complexities
- The need to register certain medical devices in Kenya
- A requirement for localised support in managing imports, warehousing, and last-mile distribution
To meet these challenges, it was essential to secure the help of a trusted logistics and regulatory partner capable of navigating complex cross-border issues, ensuring compliance with local regulations, and facilitating efficient distribution across multiple markets. They reached out to Oximio to explore a strategic partnership that could streamline their operations, support regulatory compliance, and provide scalable logistical solutions. This vital collaboration would serve as a key pillar in their growth strategy across sub-Saharan Africa.
Why Partner with Oximio?
Oximio’s comprehensive suite of services made it the ideal partner for our customer. Our strategic presence in East Africa, coupled with our deep regulatory knowledge and proven expertise in healthcare logistics, positioned us as the perfect solution provider for their needs. Our customer was impressed with the professionalism of Oximio’s operational and project management teams and our ability to solve complex logistical and regulatory challenges to global standards.
The Solution
Oximio provided a suite of tailored services that directly addressed our customer’s unique challenges. These included:
- Local Technical Representative (LTR) Services:
Acting as the company’s regulatory representative in Kenya, Oximio took charge of managing the product registration process, ensuring full compliance with the Kenyan Pharmacy and Poisons Board. This allowed our customer to focus on expanding their product offerings while we handled the intricacies of regulatory approval. - Bonded Warehouse Services:
We provided bonded warehousing, allowing our customer to import products in bulk without incurring taxes upfront. Taxes are only paid when products are released for local use, which improved cash flow and operational efficiency. This also supported the customer’s goal of using Kenya as a regional distribution hub for the wider Sub-Saharan market. - Cross-Border Transit Logistics:
Oximio offered streamlined cross-border logistics, managing shipments directly from the customer’s depot in South Africa to their customers in Kenya. We handled all aspects of import documentation, transportation, and customs clearance, ensuring timely delivery without costly delays. - Scalable Storage Solutions:
As the demand for products grew, we were able to provide scalable storage options that allowed our customer to store large quantities of ophthalmic devices efficiently. This flexibility enabled them to break bulk and distribute products quickly to various clients across Kenya. - Importer of Record (IOR) Services:
Oximio took responsibility for all import documentation, ensuring seamless customs clearance and compliance with all import regulations. This allowed the customer to operate with minimal friction and focus on their core business.
The Outcome: Growth & Operational Efficiency
The strategic partnership with Oximio has led to tangible benefits for our customer:
- Regulatory Compliance in Kenya: Our expertise in navigating the regulatory landscape helped the company achieve a solid foothold in Kenya, enabling them to expand their product offerings with confidence.
- Streamlined Cross-Border Distribution: Efficient logistics solutions significantly reduced delays and costs associated with cross-border transportation and customs clearance.
- Cost Savings and Efficiency: By leveraging Oximio’s bonded warehouse services and import management, the company was able to minimise upfront costs and reduce customs delays, improving overall operational efficiency.
- Regional Expansion: The customer is now well-positioned to grow its presence across East and sub-Saharan Africa, enhancing brand visibility and strengthening relationships with clients in multiple markets.
This partnership is a clear demonstration of Oximio’s commitment to providing scalable, tailored and compliant logistics solutions that support the growth and success of healthcare companies across the continent. Through our expertise in logistics, regulatory compliance, and warehousing, we have helped our customer not only meet their immediate operational needs but also set the stage for long-term success in the region.
Oximio in Africa
To find out how our end-to-end supply chain solutions can support your clinical trials within Africa, explore our full range of clinical trial logistic services within the region.
With a network of wholly owned and partner depots, we are able to further support your clinical research from Africa – and into Europe, North America, LATAM, MENA and Asia.
Why not contact us today to discuss your requirements. We’d be delighted to partner with you on your next clinical trial.
accum volupta nobis min porae estinve llaborehente consedit.
Supporting A Young Patient with Critical Needs in Ukraine
Introduction
At OXIMIO, our core values of care and commitment extend far beyond business – they define who we are and our mission to serve and support communities worldwide. As a leading supply chain solutions provider for clinical trials, we are dedicated to facilitating patient access to life-saving treatments. In this case study we illustrate how our team in Ukraine responded to a unique and urgent request for support, ensuring the timely importation and delivery of essential medical kits to a child suffering from spinal muscular atrophy (SMA).
Requirement
The situation began when a patient support organisation reached out to OXIMIO for assistance. The organisation was working with a child in Ukraine who had been enrolled in a 2017 multi-centre study led by a European multinational healthcare company. This study had been set up to investigate a treatment for infants with SMA Type 1, a rare and debilitating condition.
While the clinical trial had concluded, the pharmaceutical company made a five-year commitment to continue providing the child with vital medical supplies. The challenge, however, arose when the patient support organisation encountered significant logistical and regulatory obstacles in importing and delivering these life-sustaining kits. The absence of a registered post-trial programme in Ukraine complicated the process, leaving the organisation in need of a trusted partner who could navigate these complexities.
Solution
OXIMIO was uniquely positioned to address this challenge, thanks to our extensive expertise and international presence in the supply chain sector for clinical trials. Our teams in Ukraine, Hungary, and CARE collaborated closely to develop a tailored solution that identified and leveraged alternative, compliant legal pathways to address regulatory complexities and ensure the timely delivery of the required medical kits.
This success reflects the strong coordination and shared commitment of our cross-country and cross-functional teams, who worked together to design and implement a unified strategy that addressed both logistical and regulatory complexities. It included:
- Coordinating with local authorities to understand the specific regulatory requirements.
- Managing international logistics across multiple countries to facilitate the safe and efficient transport of the medical kits.
- Providing updates and continuous communication with the patient support organisation to ensure transparency and minimise any delays.
This collaborative approach allowed us to address the logistical and regulatory challenges swiftly and successfully, ensuring that the child received the necessary medical kits without disruption.
Outcome and Impact
The successful delivery of the medical kits was a testament to the power of cross-border collaboration and our ability to respond swiftly to urgent, life-changing needs. By overcoming logistical challenges and ensuring the timely importation of essential supplies, we not only supported the continuation of the child’s treatment but also reinforced our commitment to being more than just a service provider.
This initiative demonstrated our dedication to making a tangible impact on patients’ lives, especially in critical situations where time is of the essence.
Conclusion
At OXIMIO, our core values of care and commitment guide every decision we make, and this case study exemplifies our dedication to supporting patients in their most vulnerable moments. We extend our heartfelt gratitude to the patient support organisation for their trust and collaboration. Moreover, we commend our teams for their dedication, expertise, and seamless coordination across borders.
Supply Chain Solutions for Clinical Trials
To find out how we can support your clinical trial, why not explore our supply chain solutions across logistics, patient care and comparator sourcing.
Постачання необхідних гемостатичних засобів зі США до України
Гемостатичні засоби відіграють критично важливу роль у військових умовах, особливо під час бойових дій, коли виникають тяжкі кровотечі. Ці препарати є життєво необхідними в перші хвилини після поранення, оскільки забезпечують швидку зупинку кровотечі та запобігають розвитку геморагічного шоку, рятуючи життя на полі бою.
Потреба
Нещодавно до Oximio в Україні звернувся американський виробник гемостатичних засобів, який прагнув підтримати зусилля України у війні з Росією шляхом надання необхідної гуманітарної допомоги. Компанії був потрібен надійний логістичний партнер для організації імпорту та розподілу цих критично важливих медичних засобів до закладів охорони здоров’я, що забезпечують медичну допомогу на передовій.
Рішення
Маючи понад 20 років досвіду у сфері міжнародної логістики, Oximio задіяла свою розвинену мережу в Україні, включно з тісною співпрацею з Міністерством охорони здоров’я, та створила робочу групу для координації процесів імпорту, митного оформлення й транспортування близько 10 партій життєво важливих постачань.
Глибоке розуміння як міжнародних, так і місцевих логістичних процесів, особливо в умовах воєнного часу, а також регуляторних вимог дозволило забезпечити своєчасну доставку продукції з повним дотриманням встановлених стандартів якості.
Результат
Окрім успішного постачання вантажів, американська компанія здобула перемогу в тендері на надання масштабної допомоги Україні, що дозволило суттєво розширити обсяги її підтримки. Додатково було ініційовано збір даних щодо використання цих препаратів.
Завдяки ефективному та гнучкому підходу Oximio виробник зі США зміг забезпечити доставку критично важливих гемостатичних засобів до прифронтових зон України, що зробило вагомий внесок у підтримку військової медицини. Своєчасні та успішні поставки безпосередньо сприяли реалізації масштабної програми допомоги в Україні. Крім того, комплексний збір даних про застосування продукції підтримав подальші наукові дослідження та розширив знання медичної спільноти щодо ефективності гемостатичних засобів.
Висновок
Цей проект підкреслює важливість надійного, клієнтоорієнтованого логістичного партнера для забезпечення своєчасного постачання критично важливих ресурсів у складних та ризикованих умовах. Завдяки експертності та індивідуально розробленим рішенням ми не лише підтримали медичну систему України, а й зробили внесок у розвиток наукових досліджень, підтверджуючи нашу відданість зміцненню глобальної системи охорони здоров’я.
Рішення Oximio для клінічних досліджень в Україні
Ознайомтеся з нашими комплексними рішеннями у сфері управління ланцюгом постачання для підтримки клінічних досліджень в Україні або зв’яжіться з нами безпосередньо для отримання додаткової інформації. Ми будемо раді обговорити ваші потреби.
Oximio Ukraine: Leading the Way in Clinical Trial Supply Chain Solutions During the War
Despite the ongoing war in Ukraine, the need for clinical trials remains critical. In 2024 alone, from January to October, 46 new studies received approval, with 40 of them being multinational collaborations and six local initiatives. This resilience highlights the essential role of clinical research in advancing healthcare.
Established in 2004, Oximio, Ukraine has a profound understanding of patient care and the complexities of the clinical trial landscape. We are proud to have positioned ourselves as a leader in the industry, currently supporting over 62% of the new studies approved this year. Our success is rooted in our commitment to continuity and adaptability, especially in these challenging times.
Navigating Challenges with Robust Planning
Thanks to comprehensive business continuity plans that ensure uninterrupted operations and the safety of our personnel we have continued to provide agile logistics solutions for sponsors and CROs within the country. Key strategies include:
Availability and Security of Warehouse Staff
The safety and availability of our warehouse personnel are paramount. We have constructed an anti-attack shelter adjacent to our warehouse to protect our team and ensure they can carry out their daily tasks without disruption.
Reliable Power Supplies
Our facilities are equipped with two high-power diesel generators that maintain electricity in all temperature-controlled areas, including refrigerators and freezers. Regular maintenance and testing have ensured these systems function without issues, and we maintain an adequate fuel supply for uninterrupted operations.
Backup Heating Solutions
To safeguard the integrity of our temperature-sensitive materials, our depots are fitted with alternative heating solutions that operate independently of gas supplies. The ventilation system at our Krushynka depot has been modernized, and we have implemented an additional independent electric heating system connected to our generators. Meanwhile, our Brody depot features a solid fuel heating system, ensuring we can maintain optimal conditions year-round.
Distribution Availability
We have secured ongoing fuel supplies through contracts with leading gas stations and have established a three-week fuel storage supporting the uninterupted completion of all deliveries.
Robust Communication Systems
To ensure seamless communication and coordination, our action plan includes provisions for internet connectivity disruptions. We have established connections with two internet providers, alongside backup satellite and fiber optic systems, ensuring we stay connected with our clients and partners globally.
A Commitment to Patient Care
At Oximio, our core values revolve around the safety of our staff and the delivery of patient care. We understand that the stakes are high in clinical trials, and our commitment to excellence drives us to adapt and innovate continuously. The ongoing conflict in Ukraine has posed many challenges, but it has also reinforced our resolve to support vital clinical research initiatives.
As we look to the future, we remain dedicated to providing unparalleled supply chain solutions for clinical trials, ensuring that the essential work of advancing healthcare continues, even in the face of adversity. Our international team, combined with our robust operational strategies, position us as a trusted partner in the global clinical research community.
Together, we are navigating these challenging times with resilience and purpose, ensuring that patient care remains at the heart of everything we do.
Clinical Trial Supply Chain Solutions Ukraine
If you are seeking support for your clinical trial within Ukraine, view our comprehensive supply chain solutions or contact us. Offering agile and tailor-made solutions, we would welcome the opportunity to discuss your unique requirements.
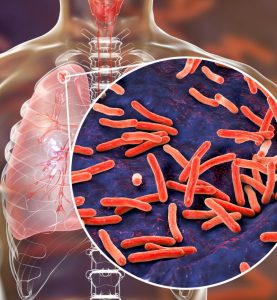
Case Study: Efficient Import, Storage, and Distribution of Diagnostic TB Kits for Research Study in Sub-Saharan Africa
Background & Challenge
Oximio recently received a call from a team of researchers in sub-Saharan Africa in urgent need of help for the import, storage, and distribution of diagnostic kits and medical devices to multiple study sites across the region. They were undertaking a study on tuberculosis (TB) which aimed to improve patient diagnostics in underserved areas. The trial involved sensitive diagnostic kits with strict temperature control ranging from frozen to ultra-low temperatures.
The challenge was to ensure that the diagnostic kits arrived at four study sites in Tanzania and Uganda in a timely manner, within the required temperature ranges, and ready for immediate use in patient diagnostics. Additionally, managing the customs processes and logistics across multiple countries, while maintaining regulatory compliance and minimising delays, was critical to the study’s success.
Solution: Streamlined Logistics and Temperature Control
To meet the demanding logistics requirements Oximio devised an efficient strategy that addressed both the regulatory complexities and the need for strict temperature control during storage and transit.
- Importation and Customs Clearance: Oximio coordinated the Import of Record (IoR) and cleared three bulk shipments through Jomo Kenyatta International Airport (NBO) in Nairobi, Kenya utilising our customs bonded warehouse near Nairobi to receive and store the shipments securely. The strategic location of our warehouse meant it was well placed to support the subsequent distribution to the study sites.
- Storage and Temperature Control: The diagnostic kits required careful storage under specific temperature conditions, ranging from -20°C to -80°C, in addition to dry ice replenishment. Oximio ensured that the equipment was stored in the appropriate temperature-controlled environments within the bonded warehouse. To maintain the integrity of the kits, we partnered with a local carrier specialising in temperature-sensitive goods for regional distribution.
- Regional Distribution: Acting as the Exporter of Record (EoR), we facilitated the distribution of 12 outbound shipments of diagnostic kits to four research sites in Tanzania and Uganda. This included handling the IOR for the goods into the receiving countries and ensuring that each shipment was appropriately packaged for transport at the required temperatures, with dry ice replenishment when necessary.
- Rapid Delivery: Oximio’s efficient logistics network ensured that the diagnostic kits reached the study sites within 72 hours—substantially reducing the standard delivery time from 6 days (if shipped directly from the UK) to just 3 days.
- Swift Regulatory Compliance: The complex process of securing Importation of Record (IoR) permits for each country was conducted in 7 days, half of the usual 14+ days it would normally take. This expedited the entire logistics process, avoiding any unnecessary delays.
Results
The complete logistics process—from importation to final delivery—was executed smoothly. The shipments arrived at the designated study sites in Tanzania and Uganda on time and within the required temperature range, ensuring the diagnostic kits were ready for use in patient diagnostics without compromising their efficacy.
The research team was able to begin their diagnostic procedures as planned, avoiding any disruptions to their study timeline. Additionally, Oximio is now poised to handle site returns and perform reconciliation and destruction of expired or used kits in compliance with the study’s protocols.
Conclusion
Oximio successfully navigated the complexities of international shipping, temperature-sensitive transport, and regulatory compliance to deliver critical diagnostic kits for a TB research study in sub-Saharan Africa. The use of our customs bonded warehouse for storage and regional distribution not only streamlined the process but also saved valuable time, ensuring that the study’s diagnostic efforts were not hindered by logistics challenges.
This case highlights the importance of local knowledge, efficient customs handling, and temperature-controlled logistics in supporting global health initiatives in remote and challenging environments. Through careful planning and strategic partnerships, we were able to deliver results that directly supported the health and research outcomes of the TB study, ensuring timely diagnostics and contributing to the fight against tuberculosis in sub-Saharan Africa.
Oximio sub-Saharan Africa
For further information on our clinical trial supply chain solutions in sub-Saharan Africa, please contact us. We’d be delighted to discuss your project requirements.
Clinical trials in Ukraine: a first-hand perspective
In this update on conducting clinical trials in Ukraine during the war we review:
A country under martial law: operating during the war and what this means for clinical trials
The impact of the war on the clinical trials market: data and anecdotal evidence
Oximio in Ukraine: our people, operations and the services we can solution
Working in partnership to prioritise patient care: Adaptability of service, prioritising shipments, factors influencing cost to serve
A case study: How alternative solutioning has enabled patients to continue on trials
The logistics of extending the footprint of advanced therapy trials
Cell and gene therapy innovators are scaling up and out to reach more patients. Clinical Trials Arena spoke to the team at Oximio (formerly the SMO Group) to discuss market developments.
Most advanced therapy medicinal products (ATMPs, also known as cell and gene therapies) address the clinical needs of “orphan” diseases (very rare conditions, where patients are so thinly spread globally that in some markets it becomes difficult or uneconomical to deliver treatments). Because of this, there are advantages to running trials across additional markets, providing access to more of the right patients.
ATMP studies have typically been designed to bring patients to a designated dosing facility, meaning they were likely to have treatment in the country where a clinical trial was running, close to where the drug was manufactured. However, therapy innovators are increasingly considering how to scale-up, run multi-country trials and bring therapies to patients, giving the opportunity for a wider group to access treatment.
Focus on geography
The US continues to be the industry front runner, with nearly half (550) of all 1,220 global ATMP trials ongoing in 2020[1]. Studies initiated in Europe have remained flat year-on-year, from 2014 to 2018, but we see evidence of the emergence of multi-country trials in the region[2].
Highly complex logistics
A major factor for selecting a clinical trial site or country are the expertise and skills of the clinical centers and healthcare professionals, followed closely by the speed of approval by regulatory authorities in country.
Logistics planning and stakeholder coordination is also a critical factor in ensuring the success of advanced therapy trials. ATMP supply chains are complex, and logistics is the element that is relatively immature, given the diversity of ATMP starting materials and the final product.
ATMPs have a variety of manufacturing strategies (centralized vs decentralized), various product characteristics (autologous vs allogeneic, fresh versus frozen) and different clinical utilizations (out-patient vs in-patient, surgical vs non-surgical). This means that bespoke logistics schemes must be created, considering specific drug characteristics, geographical coverage and the regulatory landscape.
Case study: Bespoke ATMP supply chain in action
The challenge
A developer created an allogenic cell therapy to be shipped fresh from the manufacturing site in in the EU to clinical centers in Israel. Timing was the main challenge – temperature-controlled shipping (15-25°C) for the product, across the ocean and into a patient within 72 hours. To add to the complexity, the administration was not an infusion, but surgery.
Documentation requirements were stringent too and included:
- Certificate of analysis (CoA) – to acknowledge the quality assurance for each batch produced
- Drug pedigree documents – to account for the product as it passes from manufacturer through to the eventual user
A QP release would normally be evaluated post manufacture, but ahead of the trial. The product could be held in safe controlled storage while the evaluation was completed, but that wasn’t possible with a 72-hour shelf life.
There were number of supply chain stakeholders too; the manufacturer in the EU, international transport with import and customs teams, the local delivery teams, those receiving the goods, the pharmacy and the dispensing personnel, plus a team of doctors ready to perform surgery, all of whom needed to play their part within a 36 hour window.
According to Samuel Gal Alterovich, director of strategic services and alliances at Oximio, their role was to act as a “professional integrator” between these parties. “You need a strong team of people who are knowledgeable about the challenges of clinical trial regulations in general and then specifically in the market concerned (in this case, Israel),” Alterovich explains. “But you also need someone knowledgeable about logistics, who is also able to advise on the pharmacy side and be able to prove cold chain GMP-compliant activity.”
The solution
Oximio drafted a clear standard operating procedure including the manufacturer, the global courier, the CRO and the local medical team. “We came up with a protocol in which the roles of each of the parties was very clearly articulated and so it was very clear who needed to do what and when. It all had to go like clockwork,” says Alterovich.
To make it possible, normally sequential processes had to happen in parallel. As soon as the drug was manufactured, the QP and pharmacist were ready to receive the CoA. They reviewed it while the product was on its way. And those documents – CoA and pedigree – were reviewed in real time, so any problems could be communicated, and action taken.
Upon arrival in Israel, a dedicated import team made sure the shipment was covered, that authorities were aware of it, and that the release needed to be hassle-free.
Then came the last-mile delivery, where the Oximio team was on hand to work with the local clinicians to create a GMP-like environment in the medical facility.
The result
Oximio developed tightly focused schemes to ensure that the product reached the patient in the same condition it reached the manufacturing facility, resulting in nearly 30 treatments delivered safely and on time to patients in Israel, with many more expected as the project continues.
Extending the footprint of advanced therapy logistics trials
Oximio’s capability to extend ATMP trials means partners benefit from local expertise and knowledge, including that of regulatory frameworks and culture, to facilitate improved patient access. This ability to interact with the key players in a wide range of territories, including regulators, highlights one of Oximio’s strengths – its network of local market experts. This, combined with dedicated project management in every market, makes Oximio well placed to support ATMP trials in established locations, as well as across a network of smaller, less developed markets.
Whether it’s bringing patients to advanced therapies, or bringing therapies to patients – through the establishment of additional dosing centers and communication with regulatory authorities, to the training of site staff, as well as support at import, export, domestic distribution, storage, and arrival stages – Oximio can build supply chain strategies for the success of your ATMP trial.
[1] The Alliance for Regenerative Medicine, accessed 02/09/21: http://alliancerm.org/wp-content/uploads/2021/03/ARM_AR2020_FINAL-PDF.pdf
[2] The Alliance for Regenerative Medicine, accessed 02/09/21: http://alliancerm.org/wp-content/uploads/2019/10/Trends-in-Clinical-Trials-2019-Final_Digital.pdf
Oximio’s Rapid Response: Vital Medicine Delivered from US to Africa
The need to bridge the gap between essential medicines and their accessibility can mean the difference between success and failure for any clinical trial. This was the problem that faced a local medical facility in South Africa when multiple pallets of vital supplies needed door to door delivery from the US at record speed.
After a cry for help, the logistics team at Oximio, Africa, jumped into action. Liaising with their freight contractor, they were quickly able to negotiate a fee, within a grant funded budget, for their client for airfreight, screening and airline handling. Oximio swiftly arranged collection of 17 pallets from the US supplier depot and within 24 hours, port health authorities had inspected and approved the goods for shipping on an express flight the next day. A controlled room temperature shipment, on arrival in Africa, our teams were waiting to collect and deliver the goods in record time to our customer’s premises. The whole process from start to finish took 5 days.
A job well done; the timely delivery of the medicine was greatly appreciated by our customer. This meant that the progression of their trial could continue as planned with patients receiving their essential treatment without disruption.
A huge thanks to everyone involved.
Oximio’s Clinical Trial Services
With dedicated project managers, bonded depots and warehouses across the globe, find out how we can support you with your clinical trials. Simply contact us. We’re here to help.