Clinical Trial Solutions
Caring for patients needs with patient focused solutions to enhance recruitment, retention, and engagement throughout the entirety of the clinical trial
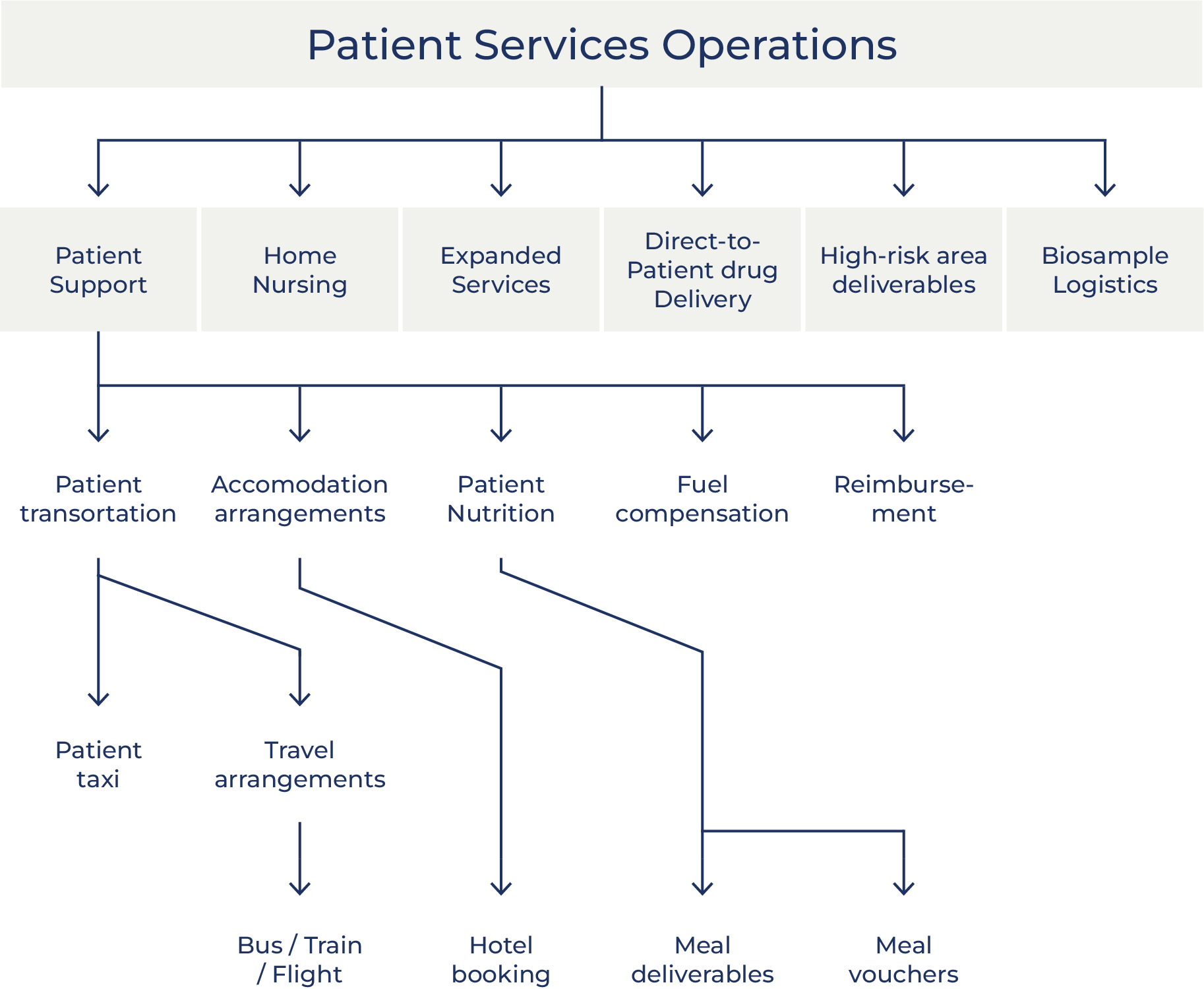
Oximio´s Patient Solutions
01.
Project Management
Project Management
Effective clinical trials project management is crucial for ensuring the successful and timely completion of research studies. We understand that navigating the complexities of clinical trials can be challenging and requires expert industry knowledge. With a variety of procedures, regulations and language barriers to deal with, our team of experienced project managers will provide comprehensive support through every stage of the process.
A clinical trial plan that’s right for you
Running a clinical trial requires stringent planning, organisation and co-ordination. A smooth supply chain delivered by solid project management is essential throughout to guarantee the smooth running of the trial. Our clinical trial project management service is designed to give you the best value for money, with flexible pricing.
View the options below and choose a plan that meets your budget.
Set-up:
for a tailored solution when needed
Management:
client needs
Licenses:
Clearance support:
Import support:
Export support:
Closure:
02.
Patient Support: Transportation
03.
Patient Support: Accommodation Arrangements
04.
Patient Support: Nutrition
05.
Patient Support: Fuel Compensation
06.
Patient Support: Patient Reimbursement
07.
Patient Support: Home Nursing
08.
Expanded Access Services, Compassionate Use & Post-Trial Access
09.
Direct-to-Patient and Direct-from-Patient Drug Delivery
10.
Biosample Logistics
Bio-samples Management
As an established logistics provider with expertise dedicated to clinical trials, Oximio provides greater efficiency and control for the collection and return of your biological samples, while minimising safety risks.
With Oximio’s bio-sample management shipment service, you can expect:
Quick and comprehensive administrative support for document management during the export of bio-samples.
Highly reliable temperature-controlled packaging
Support for a range of temperature across ambient, refrigerated, frozen and dry ice
Door-to-door service, from site to the central lab
Solutions to overcome any adverse road or infrastructure conditions
Experts in the most efficient export routes
Regulatory and customs clearance expertise
Proven implementation of solutions during military conflict and other public emergencies
Bio-sample Logistics Workflow
- The PI completes a collection request form (the form, as well as other export document templates including a letter to customs and a proforma invoice are prepared by Oximio in advance as part of the project setup)
- Oximio collects the bio-samples from the site (including controlled packaging, temperature loggers and related materials – ambient; refrigerated frozen or Dry Ice)
- Oximio undertakes the export procedures
- Oximio delivers to the central lab
- Oximio provides P.O.D
Note: As the first organisation to resume operations in Ukraine since the start of the crisis, Oximio’s services offers a comprehensive solution that puts shipment integrity and staff safety first.
Our worldwide depot network
Our global network of depots and warehouses are strategically distributed across MENA, LATAM, APAC, Europe, Africa, and North America, ensuring an agile supply chain for clinical trials whilst maintaining the highest operational standards.
Oximio Regional Map
We work according to the demands, logistics and regulations of each market around the world.
To get more local information, please click on the country of your interest.
- Oximio’s Central Depots
- Oximio’s Customs Bonded Depots
- Oximio’s Partner Depots